What Is Biological Age and How Can You Influence It?

You’re 55 years old. Your neighbor is also 55. But one of you might be biologically closer to 45, while the other functions more like a 65-year-old. This isn’t science fiction—it’s the reality of biological age versus chronological age.
Biological age represents how “old” your body actually functions at the cellular and molecular level. Unlike chronological age, which simply counts the years since your birth, biological age reflects the cumulative wear and tear your cells, tissues, and organs have experienced over time. Two people born on the same day can have dramatically different biological ages based on their lifestyle, health status, and environmental exposures.
Understanding this distinction matters because biological age is closely linked to risk of age-related diseases like cardiovascular disease, type 2 diabetes, and cognitive decline. While we cannot stop the calendar from advancing, research suggests we can influence how quickly our biology ages through lifestyle choices and, in some cases, targeted nutrition and supplementation.
At Vitruvin, we focus on supporting cellular health and longevity through evidence-based approaches. This article will walk you through what science currently tells us about biological aging, how it’s measured, what drives it, and most importantly—what you can do about it.
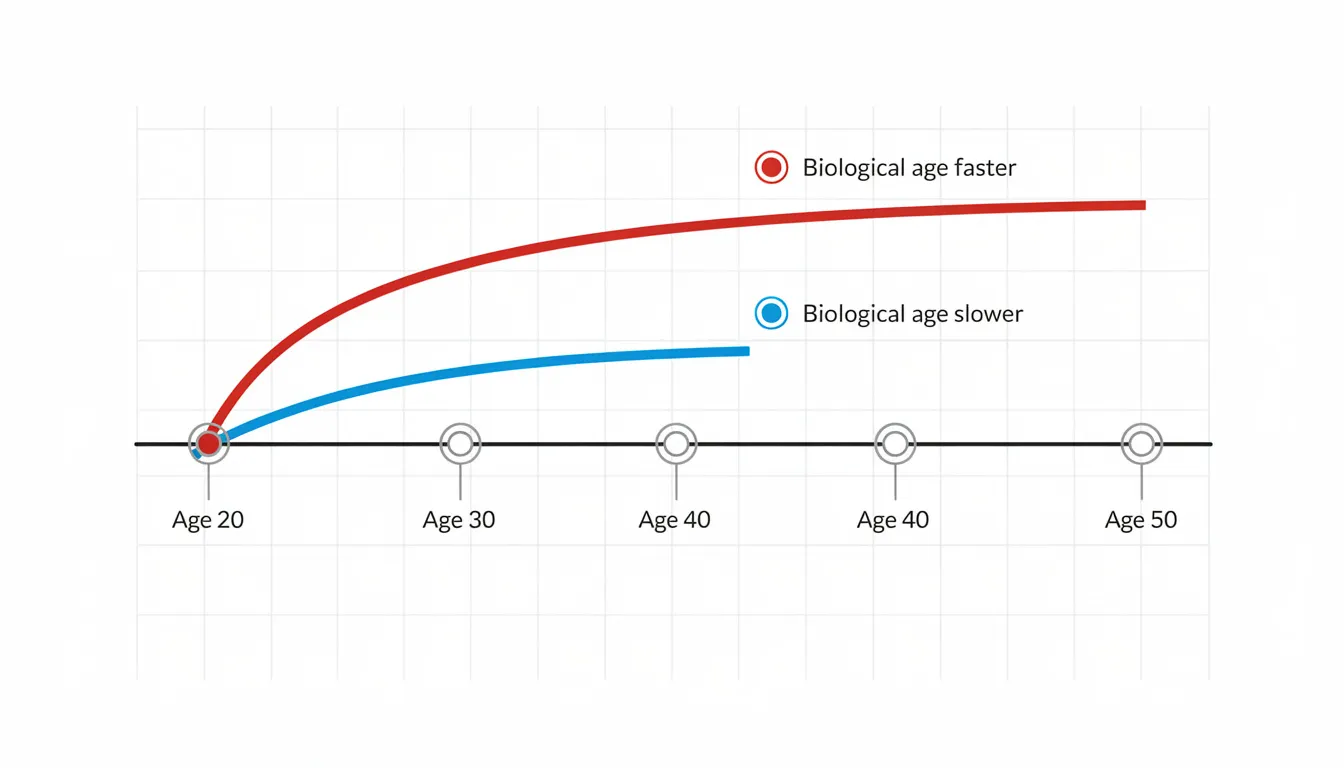
Biological Age vs Chronological Age
Let’s start with the basics. Chronological age is straightforward—it’s the number of years that have passed since your birth. Everyone ages chronologically at exactly the same rate: one year per year. No exceptions.
Biological age is different. It measures the actual wear and tear at your organ, cellular, and molecular levels. Think of it as your body’s internal odometer versus how many years the car has been on the lot.
Consider this concrete example: a 60-year-old with excellent cardiovascular fitness, normal blood pressure, good sleep patterns, and healthy eating habits may have a biological age closer to 50. Meanwhile, a 45-year-old with obesity, high blood sugar, a sedentary lifestyle, and chronic stress might function biologically like someone in their late 50s or early 60s.
This brings us to an important concept: healthspan. While lifespan measures total years lived, healthspan refers to years lived in good health, free of major disability or chronic disease. The goal isn’t just to live longer—it’s to live better for longer.
Major epidemiological studies like the Framingham Heart Study and UK Biobank have demonstrated enormous variation in health outcomes among people of the same chronological age. Some 70-year-olds run marathons while others struggle with basic mobility. This variation points to the influence of modifiable factors on biological aging.
The encouraging takeaway? Your actual age doesn’t have to dictate your health trajectory.
How Scientists Estimate Biological Age Today
As of 2026, there is no single gold-standard “biological age test.” Instead, researchers use multiple methods that capture different aspects of the aging process. Think of it like trying to assess the overall condition of a house—you’d check the foundation, roof, plumbing, and electrical separately to get a complete picture.
Scientists often combine several markers to approximate biological age:
Clinical laboratory values (blood pressure, glucose, lipids)
Functional tests (grip strength, walking speed)
Molecular signatures (DNA methylation patterns, telomere length)
Commercial biological age tests have become available through various companies offering saliva or blood-based analysis, fitness algorithms, and more. However, many of these tests are still being validated, and their results should be interpreted cautiously—ideally with guidance from a healthcare professional.
It’s worth noting that Vitruvin does not diagnose or measure biological age. We follow this research closely to inform our educational content and consultative guidance, helping people make informed decisions about supporting their cellular health.
Main Biological Mechanisms That Drive Aging
In 2013, researchers published a landmark paper identifying the “hallmarks of aging”—fundamental processes that drive biological decline. This framework was updated in 2023, reinforcing that human aging is multifactorial rather than caused by any single mechanism.
These processes occur simultaneously and influence each other:
Genomic instability: Accumulating DNA damage and mutations
Telomere shortening: Protective chromosome caps wearing down
Epigenetic alterations: Chemical changes affecting gene expression
Mitochondrial dysfunction: Reduced cellular energy production
Cellular senescence: Cells that stop dividing but remain active
Chronic inflammation: Low-grade systemic inflammation
Stem cell exhaustion: Reduced regenerative capacity
Microbiome changes: Shifts in gut bacterial composition
The empowering news? Lifestyle and environmental factors can speed up or slow down many of these mechanisms, even when genetic factors set a starting point.
Genomic Instability and DNA Damage
Genomic instability refers to the accumulation of DNA damage and mutations over time. This damage comes from two main sources:
Internal sources: Normal metabolic processes that produce reactive oxygen species
External sources: UV light, tobacco smoke, environmental toxins, and certain pollutants
Your cells have sophisticated repair systems to fix this damage. However, these repair mechanisms gradually become less efficient with age, increasing the risk of errors in cell function. This is why environmental exposures matter so much—repeated exposure to damaging agents accelerates the process.
Large-scale studies have linked higher markers of DNA damage with increased risk of age-associated conditions, including cancer and cardiovascular disease. The protective behaviors are straightforward: sun protection, avoiding tobacco smoke, limiting exposure to industrial chemicals, and maintaining overall health.
Telomere Shortening
Telomeres are the protective caps at the ends of your chromosomes, often compared to the plastic tips on shoelaces that prevent fraying. Every time your cells divide, these caps get slightly shorter.
Here’s the biological reality: most human cells can only divide a finite number of times before entering a non-dividing state. This is called the Hayflick limit, named after the researcher who discovered it. At birth, average telomere length ranges from approximately 8,000 to 15,000 base pairs. With each cell division, this number decreases.
Research suggests that shorter telomeres in white blood cells are associated with:
Higher risk of earlier mortality
Increased incidence of many chronic diseases
Greater vulnerability to age related diseases
What influences telomere length? Studies consistently associate shorter telomeres with:
Chronic stress
Smoking
Obesity
Sedentary lifestyle
Conversely, regular physical activity and a high-quality diet rich in nutrients are associated with relatively longer telomeres in population studies.
Epigenetic Alterations and DNA Methylation Clocks
Epigenetics refers to chemical changes that regulate which genes are more or less active without altering the underlying DNA sequence. Think of it like software running on hardware—the code stays the same, but which programs are running changes.
The most studied epigenetic modification involves DNA methylation patterns—methyl groups attaching to specific locations on DNA. These patterns change in fairly predictable ways with age, which has allowed researchers to build “epigenetic clocks” that estimate biological age.
The most well-known epigenetic clock was developed by Steve Horvath in 2013, analyzing methylation at over 350 CpG sites across 51 tissue types. Since then, additional clocks like Hannum, GrimAge, and DunedinPACE have been developed with different strengths.
Key findings from epigenetic aging research:
When epigenetic age exceeds chronological age (called “epigenetic age acceleration”), studies show 8-15% higher mortality risk per year of acceleration
Different organs can age at different rates—your heart might be biologically younger than your liver
Emerging pilot studies suggest lifestyle interventions may modestly influence these clocks
This remains an active area of aging research, with new clocks and validation studies published regularly.
Cellular Senescence
As cells divide and accumulate damage, some enter a state called cellular senescence—they permanently stop dividing but remain metabolically active. This isn’t inherently bad; senescent cells play beneficial roles in development and wound healing.
The problem emerges when senescent cells accumulate chronically. These cells secrete inflammatory molecules (called the senescence-associated secretory phenotype, or SASP), contributing to:
Tissue dysfunction
Chronic inflammation
Impaired organ function
Animal studies where researchers selectively removed senescent cells showed improved tissue function and extended healthy lifespan. However, these are not yet established therapies for humans—this remains an experimental frontier.
Mitochondrial Dysfunction and Energy Metabolism
Mitochondria are often called the “powerhouses” of your cells, generating the ATP that fuels nearly every cellular process. They also regulate cell death and maintain redox balance.
With age, mitochondrial health tends to decline:
Mitochondrial DNA accumulates damage
Quality control mechanisms (mitophagy) become impaired
Energy output decreases
Reactive oxygen species production increases
This decline connects to common age-associated experiences: reduced muscle endurance, slower recovery from exercise, and decreased overall vitality.
Factors associated with healthier mitochondrial function include:
Regular aerobic and resistance exercise
Sufficient quality sleep
Good metabolic health (stable blood sugar, healthy insulin sensitivity)
Stem Cell Exhaustion and Impaired Regeneration
Stem cells are specialized cells capable of generating new cells to replenish tissues—blood, skin, gut lining, muscle, and more. They’re your body’s internal repair crew.
With age, stem cells face multiple challenges:
Accumulated DNA and epigenetic damage
Exposure to inflammatory signals
Hostile cellular environments
The result? Reduced regenerative capacity, which manifests as:
Slower wound healing
Bone loss and decreased bone density
Reduced muscle mass (sarcopenia)
Thinner, less resilient skin
Research into stem-cell-based rejuvenation is active but largely experimental. For now, the focus should remain on lifestyle measures that support overall tissue health.
Chronic Inflammation, Immune Aging, and the Gut Microbiome
“Inflammaging” describes the low-grade, chronic inflammation that tends to increase with age. This systemic inflammation is associated with higher risk of multiple chronic diseases, from cardiovascular disease to cognitive decline.
The immune response itself ages—a process called immunosenescence. This leads to:
Weaker responses to new infections and vaccines
Increased autoimmunity in older adults
Reduced ability to clear damaged cells
The gut microbiome plays a crucial role here. As we age, microbial diversity often decreases, with more pro-inflammatory species appearing—particularly in frail older women and men.
Cohort studies show that dietary patterns commonly found in healthy aging populations—those rich in fiber, polyphenols, and minimally processed foods—are associated with more favorable microbiome profiles and lower inflammatory markers.
Key Ways Biological Age Is Estimated in Practice
When most people encounter biological age, it’s through one or more tests that reflect different body systems. Understanding these methods helps you interpret any results you might receive.
The most informative approach combines:
Clinical markers: Blood pressure, lipid panels, glucose levels
Functional capacity: Grip strength, walking speed, balance
Molecular tests: DNA methylation analysis, sometimes telomere length
No single test captures the complete picture of your overall health and aging status. And none of these tools replace medical diagnosis or individualized care—they’re risk indicators and monitoring tools.
Use any biological age estimate as motivation to adopt healthy living habits, not as a fixed label defining your future.
Clinical and Functional Biomarkers
Common clinical indicators linked to biological aging include:
Resting blood pressure and heart rate
Fasting glucose and HbA1c (blood sugar markers)
Lipid profile (cholesterol, triglycerides)
Kidney and liver function markers
Inflammatory markers like hs-CRP
Composite scores built from these metrics (like PhenoAge) have been correlated with mortality and disease risk in large cohorts.
Simple functional tests used in research and clinical practice include:
Gait speed (4-6 meters) to assess overall mobility and physical function
Grip strength as a measure of muscle strength and overall vitality
Sit-to-stand test evaluating lower body strength and balance
Balance tests to assess vestibular function and fall risk
These tests are simple, inexpensive, and surprisingly predictive of future health outcomes. Work with healthcare professionals to interpret results and track changes over time.
Telomere Length Testing
Telomere length can be measured from blood samples using methods like quantitative PCR or specialized laboratory assays. However, there are important limitations:
High inter-individual variability
Measurement error between tests
Influence of short-term factors on results
Single snapshots may not reflect long-term trends
Professional organizations currently do not recommend routine telomere testing for clinical decision-making in the general population. If you choose commercial telomere tests, treat results as rough indicators rather than precise measurements—and avoid anxiety over small differences.
Epigenetic Clocks and DNA Methylation Tests
Several commercial laboratories now offer DNA methylation-based biological age estimates using blood or saliva samples. Research epigenetic clocks have shown robust associations with aging outcomes in cohort studies.
Practical considerations include:
Cost (typically $200-500 per test)
Need for specialized analysis
Variability between different clock algorithms
Methods continue to evolve
If you consider such testing, work with qualified professionals who understand current evidence and limitations. A single number shouldn’t drive major decisions without broader context.
Composite and AI-Based Aging Clocks
Emerging approaches use artificial intelligence and machine learning to integrate multi-omics data—combining genomics, epigenomics, proteomics, and metabolomics with clinical and lifestyle data.
Studies from 2019-2024 show these deep learning models often produce more accurate age estimates than single-biomarker methods, with some achieving 95%+ accuracy in mortality prediction.
However, these tools remain mostly research-grade and aren’t yet widely accessible or standardized for individual use. Future clinical practice may use such composite scores to personalize preventive strategies, but this remains a developing field.
How Lifestyle Can Influence Biological Age
Large cohort and intervention studies consistently associate certain lifestyle patterns with slower biological aging and lower risk of age-related conditions. This is where the science becomes actionable.
A critical caveat: lifestyle is not a guarantee against disease. Genetic factors, early-life conditions, and social determinants all play major roles. Some people do everything “right” and still develop health conditions; others seem to violate every rule and live to 95.
That said, the evidence strongly supports five major domains for influencing biological age:
Physical activity
Nutrition
Sleep
Stress and mental health
Social connection
The goal isn’t perfection—it’s progress through small, sustainable changes.
Physical Activity and Muscle Strength
Regular physical activity is one of the most powerful, consistently supported levers for healthy aging. The evidence here is remarkably strong.
Current guidelines from the WHO and other health organizations recommend:
At least 150-300 minutes of moderate aerobic activity per week
Muscle-strengthening activities on 2+ days per week
Research suggests that cardiovascular fitness, as measured by VO₂ max or simple tests like the 6-minute walk, is strongly linked to mortality risk independent of calendar age. Studies on endurance athletes reveal they often have epigenetic ages 5-10 years younger than sedentary peers of the same chronological age.
What does exercise do at the cellular level?
Improves mitochondrial function and energy production
Enhances insulin sensitivity and metabolic health
Reduces markers of chronic inflammation
Supports immune function
May help maintain telomere length
Practical examples that work:
Brisk walking (30 minutes daily)
Cycling or swimming
Bodyweight exercises (squats, push-ups)
Resistance training with bands or weights
If you’re starting from a sedentary lifestyle, begin slowly and seek medical clearance if you have existing health conditions.

Nutrition Patterns and Metabolic Health
Dietary patterns associated with slower biological aging share common features:
Abundant vegetables and fruits
Whole grains and legumes
Nuts and seeds
Healthy fats (olive oil, fatty fish)
Adequate protein
Minimal ultra-processed foods
Limited added sugars and trans fats
The Mediterranean-style diet rich in these elements consistently shows benefits for cardiovascular mortality, cognitive decline prevention, and overall longevity in research.
Better diet quality scores (like the Alternate Healthy Eating Index) are linked with:
Lower epigenetic age acceleration
Reduced insulin resistance
Decreased chronic disease risk
Better microbiome diversity
Blood sugar stability matters significantly. Strategies include:
Regular meals rather than sporadic eating
Minimally processed carbohydrates
Protein with each meal
Mindful portion sizes
Limiting sugary drinks and refined grains
Some individuals benefit from professional guidance—especially those managing diabetes, kidney disease, or other specific health conditions.
Sleep Quality and Circadian Rhythms
Chronic sleep restriction and irregular schedules are associated with:
Higher inflammation markers
Altered hormone balance
Increased risk of premature aging
Metabolic dysfunction
Research links short or fragmented sleep with higher risk of cardiovascular disease, obesity, and blood sugar dysregulation.
Most adults should aim for 7-9 hours of sleep per night with consistent bed and wake times, though individual variation exists.
Evidence-based sleep hygiene includes:
Reduce evening screen exposure (blue light suppresses melatonin)
Keep bedroom dark and cool (supports natural sleep temperature drop)
Limit caffeine after early afternoon (caffeine has 6+ hour half-life)
Avoid heavy meals close to bedtime (digestion can disrupt sleep)
Maintain consistent sleep schedule (reinforces circadian rhythm)
Stress, Mental Health, and Resilience
Chronic psychological stress impacts biological age through multiple pathways. Studies of caregivers and trauma-exposed populations show associations with:
Shorter telomeres
Higher epigenetic age
Elevated inflammatory markers
Impaired immune response
Acute stress is natural and even adaptive. The problem is prolonged, unrelieved stress that keeps the body in a chronic fight-or-flight state.
Practical strategies with evidence for stress reduction:
Mindfulness-based stress reduction programs
Regular moderate physical activity
Cognitive-behavioral approaches
Meaningful hobbies and creative pursuits
Structured problem-solving techniques
Professional support from therapists, counselors, or psychiatrists should be sought for ongoing anxiety, depression, or trauma-related symptoms. Mental health is integral to healthy living.

Social Connection and Purpose
Strong social relationships are linked to lower mortality risk—comparable in magnitude to some traditional risk factors like smoking and physical inactivity.
Research shows that social isolation and loneliness are associated with:
Higher biological age estimates
Increased incidence of cardiovascular events
Accelerated cognitive aging
Greater overall mortality risk
The quality of relationships matters as much as quantity. Meaningful connections with family, friends, and community provide psychological resilience and practical support.
Realistic suggestions for strengthening social connection:
Join interest groups or clubs
Engage in local community activities
Volunteer for causes you care about
Maintain regular check-ins with friends and relatives
Consider group fitness classes for combined social and physical benefits
Evidence-Based Approaches That May Support Cellular Health
This section summarizes current research on nutrients and compounds studied in the context of aging biology. These are not cures or guaranteed age-reversing interventions—the evidence continues to evolve.
Important: Consult healthcare professionals before starting any supplement, especially if you take medications or have medical conditions.
Research has explored several classes of compounds:
NAD⁺ precursors (NR, NMN)
Polyphenols (resveratrol, quercetin)
Autophagy-supporting nutrients (spermidine)
Collagen and joint-supportive nutrients
Cognitive support compounds (lion’s mane mushroom)
NAD⁺ Metabolism and Precursors
NAD⁺ (nicotinamide adenine dinucleotide) is central to cellular energy production and DNA repair. Research in animal models and some human tissues shows NAD⁺ levels decline with age.
Precursors like nicotinamide riboside (NR) and nicotinamide mononucleotide (NMN) have been studied for their ability to boost NAD⁺ levels. Animal studies show effects on mitochondrial function and metabolic health.
Early human clinical trials report:
Changes in blood NAD⁺ levels
Some improvements in metabolic parameters
Generally good safety profiles at studied doses
However, long-term safety data and clinical outcome data (cardiovascular events, lifespan effects) remain limited as of 2026. NAD⁺-related supplementation should be viewed as an adjunct to, not substitute for, core lifestyle measures.
Discuss potential interactions with clinicians, particularly if you have cancer history, liver disease, or take multiple medications.
Polyphenols and Mitochondrial Signaling (e.g., Resveratrol)
Resveratrol is a polyphenol found in grapes, berries, and peanuts. It’s been widely studied in animal models for effects on stress resistance pathways and mitochondrial function.
The evidence picture:
Rodent data are promising
Human evidence is mixed, often based on small studies
Most human trials use surrogate markers rather than hard outcomes
Dietary polyphenols from whole foods—berries, colorful vegetables, olive oil, tea—are consistently associated with better cardiovascular and metabolic profiles in observational research.
The practical approach: prioritize polyphenol-rich diets first, and consider supplements only under guidance with realistic expectations.
Autophagy and Cellular Cleanup (e.g., Spermidine)
Autophagy is your cell’s recycling and cleanup system, clearing damaged proteins and organelles to maintain function. Think of it as cellular housekeeping.
Spermidine is a naturally occurring polyamine found in:
Soy products
Mushrooms
Whole grains
Aged cheese
Legumes
European cohort studies have linked higher spermidine intake with reduced mortality, though causality isn’t firmly established. The National Institute on Aging and other research bodies continue investigating these relationships.
Emphasize food sources and overall dietary patterns rather than relying solely on isolated compounds.
Collagen, Connective Tissues, and Musculoskeletal Aging
Collagen is a major structural protein in skin, cartilage, tendons, and bone. Collagen turnover and cross-linking patterns change with age, contributing to wrinkles, joint stiffness, and reduced tissue resilience.
Both marine and bovine collagen supplements have been evaluated in randomized trials for:
Joint comfort
Skin hydration and elasticity
Muscle mass preservation in older adults
Results show modest positive effects in some studies. However, supporting endogenous collagen maintenance requires:
Adequate total protein intake
Vitamin C (essential for collagen synthesis)
Regular strength training
Collagen may support specific tissues but shouldn’t be framed as a systemic “anti-aging” cure.
Compounds Studied for Cognitive Support (e.g., Lion’s Mane)
Lion’s mane (Hericium erinaceus) is a medicinal mushroom traditionally used in East Asia, now studied for potential effects on nerve growth factors and cognition.
Small randomized trials in older adults with mild cognitive complaints show:
Modest improvements in some cognitive tests
Generally favorable safety profiles
Limitations include small sample sizes and short study durations.
Lifestyle factors remain the foundation for supporting cognitive health:
Physical activity
Quality sleep
Cognitive engagement and learning
Blood pressure management
Metabolic health maintenance
Any use of cognitive-support supplements should be discussed with a clinician, especially when other medications are involved. Avoiding premature aging of the brain requires a comprehensive approach.
How to Start Influencing Your Biological Age in the Next 30 Days
Theory is valuable, but action creates change. Here’s a practical 30-day framework focused on small, achievable habits rather than dramatic overhauls.
The approach: pick 1-2 goals from each domain and track adherence—not perfection.
Movement
Daily 20-30 minute walk
2-3 strength sessions per week (even 15 minutes counts)
Nutrition
Add one extra serving of vegetables daily
Include protein with every meal
Reduce ultra-processed snacks
Sleep
Set a consistent bedtime
Create a 30-minute wind-down routine
Stress
5 minutes of daily breathing practice
Weekly time for a meaningful hobby
Social
Schedule one social interaction per week
Reach out to one friend or family member daily
Simple tracking tools work best: a paper habit tracker, smartphone notes, or basic wearable data for steps and sleep duration.
Building a Realistic Personal Plan
Start by honestly assessing your current baseline:
How active are you really?
What does a typical day of eating look like?
How much sleep are you actually getting?
What’s your stress level on a 1-10 scale?
When did you last have meaningful social contact?
Set SMART goals for your 30-day experiment:
Specific: “Walk for 20 minutes after dinner” vs “exercise more”
Measurable: Track completion daily
Achievable: Start with what’s realistic given your current fitness
Relevant: Choose changes that matter to you personally
Time-bound: Commit to 30 days initially
Start with the easiest high-impact change to build confidence. Walking after dinner or adding breakfast protein requires minimal willpower but yields noticeable health benefits.
Plan for obstacles:
Bad weather? Have an indoor backup (stairs, home exercises)
Time constraints? Break exercise into 10-minute chunks
Motivation dips? Remember why you started; enlist an accountability partner
Consider collaborating with a healthcare provider or health coach for accountability and tailored adjustments based on your specific health conditions.
Monitoring Progress Without Becoming Obsessive
Pay attention to subjective changes—these often matter more than numbers:
Energy levels throughout the day
Mood stability
Sleep quality
Ease of daily physical tasks
Overall sense of well-being
For those with access to regular healthcare, track simple markers over months to years:
Blood pressure trends
Waist circumference
Basic lab tests (glucose, lipids)
Rather than chasing frequent “age scores,” focus on trajectory over time.
Warning signs of over-monitoring:
Anxiety about daily weight or step count fluctuations
Compulsive checking of health metrics
Distress when numbers aren’t “perfect”
Self-compassion and flexibility matter. Setbacks are normal. Long-term consistency matters far more than perfect adherence. The goal is an accurate measure of progress, not obsessive tracking.
Key Takeaways: What You Can and Cannot Control
Biological age reflects the cumulative state of your cells, tissues, and systems. It can diverge significantly from your chronological age—for better or worse.
What you can influence:
Physical activity and movement patterns
Diet quality and eating habits
Sleep duration and consistency
Stress management practices
Social connections and sense of purpose
Avoidance of tobacco and excessive drink alcohol habits
Informed use of supplements where appropriate
What’s harder to control:
Family history and inherited genetic factors
Early-life conditions and exposures
Some environmental exposures (air quality, historical toxin exposure)
Random biological variation
Research suggests that while genes set roughly 20-30% of the variance in biological aging, habits drive approximately 70% of your trajectory. This is empowering—your choices matter significantly.
The evidence from various aspects of aging research points to a consistent conclusion: consistent, incremental changes over years outperform any single “anti-aging” breakthrough. There is no magic pill, no secret supplement, no hidden technique that replaces the fundamentals.
While we cannot choose our chronological age, we can influence how well we live those years. Healthy aging isn’t about fighting time—it’s about supporting your biology day by day, making choices that reduce cellular damage and enhance your body’s remarkable repair mechanisms.
Start with one change. Make it stick. Add another. This is how you build a foundation for aging compared favorably to your peers and, more importantly, to your own potential.
Your biological age isn’t fixed. What will you do this week to shift it in your favor?



Comments